Unit 9 - Every breath you take
Content
- MYP10u9.1 - The atmosphere
- MYP10u9.2 - The physical properties of gases
- MYP10u9.3 - The gas laws
- MYP10u9.4 - Respiration in animals
- MYP10u9.5 - The mechanism of gas exchange
- MYP10u9.6 - Metal and non-metal oxides
- MYP10u9.7 - Atmospheric pollution
- MYP10u9.8 - The carbon cycle
- MYP10u9.9 - The rate of chemical reactions
- MYP10u9.10 - Now test yourself
Skills and understanding
- Describe gas exchange in vertebrates and invertebrates
- Predict the acid-base nature of oxides using the periodic table
- Use the gas law equations to solve simple problems
- Describe the Greenhouse effect.
- Determine the rate of gas evolution.
Scheme of work
9.1 - The atmosphere
The atmosphere is defined as the layers of gas that exist all around the Earth. The atmosphere gradually gets thinner are you go higher up.
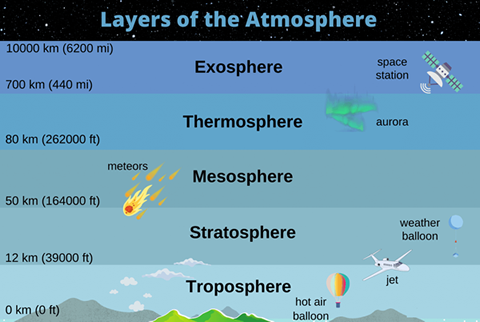
The atmosphere consists of mainly five gases, nitrogen, oxygen, argon, water vapour and carbon dioxide. There are also trace gases such as methane and ammonia.
Harmful gases that have been put into the atmosphere through human activity are known as pollutants.
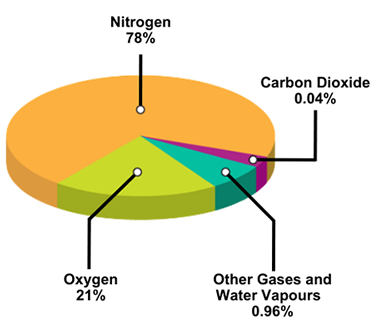
You do not need to know the exact percentages, but you should know that nitrogen occupies most of the total (4/5) and oxygen (1/5) approximately the rest. Carbon dioxide is between 0.02 and 0.04% (why would it vary?).
Check your understanding
- What are the main gases that make up the Earth's atmosphere?
- Why does the percentage of carbon dioxide in the atmosphere vary?
- What is the difference between natural gases and pollutants?
9.2 - The physical properties of gases
Like all matter, gases consist of particles. The particles have enough energy to break any forces of attraction between them.
This energy is in the form of motion - gas particles move very fast (about 1000m/s at room temperature on average). This means that gases will expand rapidly to fill all available volume.
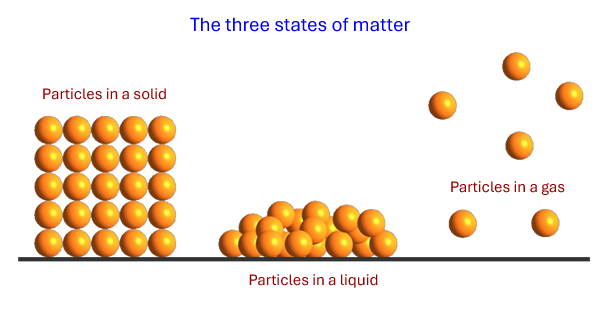
The physical properties of gases are the result of the particles being spread out and moving around very fast.
Compressible
Gases can be squeezed into smaller volumes as they are mostly empty space. They are said to be "compressible"
Diffusion
Gases spread out in all directions, as their particles are moving very fast. If there were no air in the way, a gas released into an empty volume would spread out almost instantly. As it is, gases diffuse through air at a speed that depends on the size of the gas particles.
Check your understanding
- Why are gases compressible?
- What is meant by diffusion, and how does it relate to particle motion?
- Why do gases fill the entire space available to them?
9.3 - The gas laws
The gas laws are the result of observations made on gases. They tell us how gases behave in terms of their volume, pressure and temperature.
Volume
The volume of a gas is simply the size of its container. This is because gases spread out to fill the whole container.
A cubic box of side 10cm has a total volume of 10 x 10 x 10 = 1000 cm3
Pressure
The concept of pressure is a little more complex to describe. Gas particles collide with each other and with the walls of the container. Every single collision with the walls exerts a force on the wall. The definition of pressure is the total force divided by the area on which it acts.
Pressure = force/area
Determine the pressure when a force of 100N acts on a surface of area 2 m2.
Pressure = force/area
Pressure = 100N/2m2 = 50 Nm-2
The units of pressure are the Newton per metre squared, Nm-2, which is also called the Pascal, Pa.
1 Pa = 1 Nm-2
Investigating the pressure of your footprint
The hypothesis is that boys exert more pressure on the floor than girls because they are heavier on average.
Instructions
- Trace the outline of your shoe on squared paper.
- Count the squares. Each small square is 0.25 cm2.
- Weigh yourself on the balance provided.
- Divide your mass by the surface area of your shoe in m2 (10000 cm2 = 1 m2)
- Share your data with four members of the same gender.
- Compare your five results with the results of a group of the opposite gender.
Atmospheric pressure
The pressure of the atmosphere is caused by the large column of air above our heads being pulled downwards by gravity. There are more air particles near the surface and so the pressure is greater. The atmospheric pressure at sea-level is 100 kPa (1 x 105 Pa).
As we go higher up from sea-level the pressure drops. In Madrid the air pressure is usually about 6% lower than at sea-level, the actual value also depends on the weather conditions.
Mountaineers often need to carry oxygen to breathe, as the air becomes too thin to support normal respiration.
The PhyPhox application can be downloaded onto Android devices to give a measurement of the air pressure at any time.
Air pressure measured at the top of Beret, Pyrinees
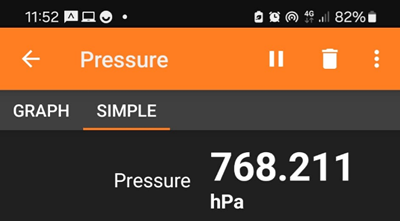
In the image the data shows measurement in hPa (hectaPascals), where 10 hPa = 1 KPa. Hence, the air pressure was 76.8 kPa, or about 25% lower than atmospheric pressure.
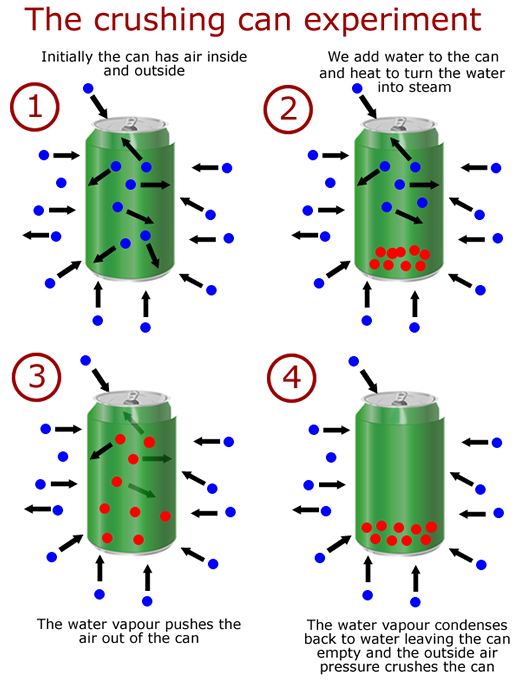
The relationship between pressure and volume
When pressure is applied to a volume of gas the volume decreases. This kind of relationship is called inverse proportionality. We say that the volume is inversely proportional to the pressure. As the pressure increases the volume decreases, and vice versa.
We can show this using a sealed gas syringe with weights. As we apply weights to the syringe plunger, the force down increases. This causes the plunger to enter the syringe further, reducing the volume.
Mathematically, the relationship is:
Volume = 1/Pressure
Explanation
The reason is that the externally applied force pushes the particles inside the container closer together. This reduces the volume making the available surface that the particles can collide with smaller, so that there are more collisions per unit area.
The relationship between pressure and temperature
If we increase the temperature of a fixed volume of gas, the pressure increases.
If we take a sealed container connected to a pressure sensor, the container has a fixed volume. As we increase the temperature the pressure inside the syringe increases. The pressure meter or sensor increases its reading.
Mathematically, the relationship is:
Pressure ∝ Temperature
Explanation
The reason is that the increase in temperature gives the particles inside the container more energy and makes their collisions with the walls of the container more energetic. Also, the particles are moving faster, so there are also more collisions with the walls of the container per unit time.
The relationship between volume and temperature
If we increase the temperature of a fixed volume of gas, the volume increases.
This can be shown using a gas syringe which is free to move to equalise the pressure inside with the atmospheric pressure. If the syring is heated the plunger moves further out, increasing the volume.
Mathematically, the relationship is:
Volume ∝ Temperature
Explanation
The reason is that increased energy as the temperature rises causes the particles to move faster on average. This increases the pressure inside the syringe, which forces the plunger to push out against atmospheric pressure until the pressure outside is equal to the pressure inside.
Check your understanding
- State the mathematical relationship between pressure and volume of a gas.
- Why does increasing the temperature of a gas increase its pressure (at constant volume)?
- What is the value of atmospheric pressure at sea level in kPa?
9.4 - Respiration in animals
Respiration is the process by which living organisms obtain energy for their life functions.
There are three fundamental methods that animals use to obtain oxygen for respiration.
- Lungs - vertebrates on land
- Gills - fish
- Diffusion - invertebrates
Living creatures use oxygen to convert glucose or other food stuffs into energy for homeostasis, growth and locomotion. The by-products (waste) of respiration in animals are carbon dioxide and water.
The actual chemical processes are quite complex, but can be summarised as:
Respiration in animals
Glucose + oxygen → carbon dioxide + water + energy
C6H12O6 + 6O2 → 6CO2 + 6H2O + energy
How lungs work
The lungs are bags of air sacs connected to the chest cavity. When the muscles cause the chest cavity to expand the volume inside the lungs increases. We have already seen that when the volume of a gas increases its pressure decreases.
With reduced pressure inside the lungs, the air pressure outside forces air into the lungs, which can then absorb the oxygen into the bloodstream for use in respiration.
Modelling lungs in the laboratory
We can use a bell jar with a rubber surface to model the operation of lungs.
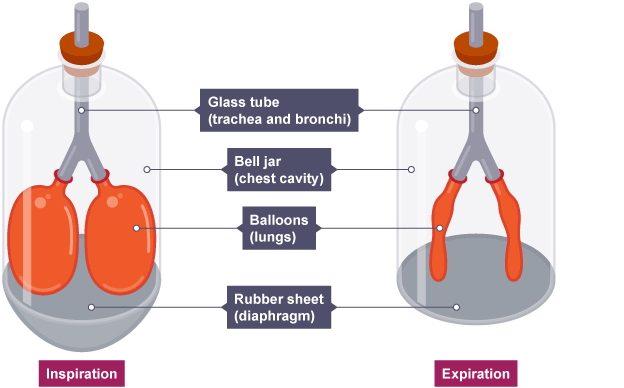
When the rubber sheet is pulled down the volume increases and the reduced pressure inside the bell jar causes the air already in the balloons to expand.
This is not a perfect model, as we are reducing the air pressure in the wrong place, but the effect is similar.
In the body system, the lungs themselves are attached to the walls of the chest and forced to increase in size by the chest muscles and diaphragm (below).
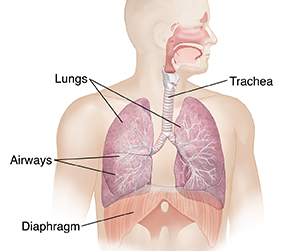
Check your understanding
- Write the word and symbol equations for aerobic respiration in animals.
- List three different ways animals obtain oxygen for respiration.
- How does increasing the volume of the lungs help air to enter them?
9.5 - The mechanism of gas exchange
Animals breathe to obtain oxygen and to remove the waste products of respiration, carbon dioxide. Just how this happens is known as the mechanism of gas exchange.
- Air enters the lungs and is drawn to the alveoli.
- The alveoli are surrounded by blood vessels.
- The oxygen diffuses into the blood where it is picked up by the red blood cells.
- At the same time, the red blood cells drop off their carbon dioxide, which diffuses out into the alveoli.
- The oxygen is transported to the cells, where it is used for respiration.
- The red blood cells pick up the waste product of respiration, the carbon dioxide and carry it back to the lungs.
The lungs
The lungs have been discussed in the previous section.
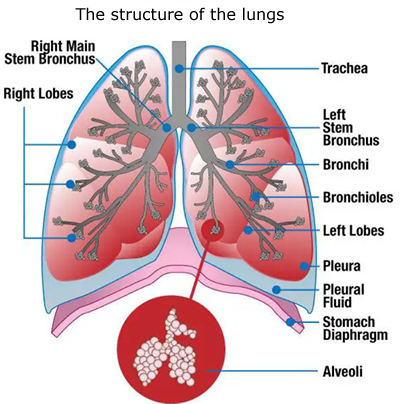
The alveoli
The alveoli (singular: alveolus) are tiny sacs at the ends of the lungs' branches and bronchioles surrounded by blood vessels. They have very large surface area to facilitate the gas exchange.
Oxygen gas leaves the air inside the alveoli and passes into the bloodstream via the blood vessels, while at the same time carbon dioxide passes from the blood stream to the air inside the alveoli.
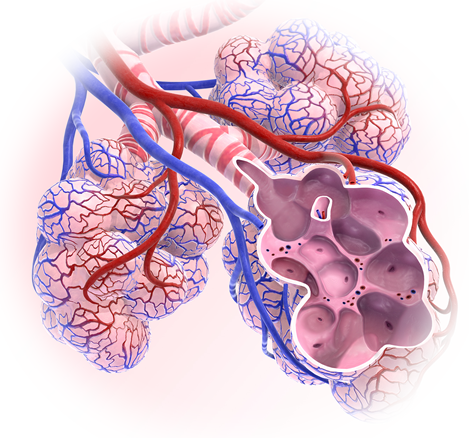
The red blood cells
The oxygen is bonded to the red blood cells using a complex molecule called haemoglobin. The blood cells then carry oxygen in the blood stream to the cells, where it is used for respiration.
Check your understanding
- Where does gas exchange take place in the lungs?
- Explain how oxygen and carbon dioxide move between alveoli and blood.
- What is the role of red blood cells in gas transport?
9.6 - Metal and non-metal oxides
Oxygen, element number 8, is a very reactive substance that can form compounds with almost every element. These compounds are called oxides.
Most oxides are very much more stable than the elements that originally made them. Carbon dioxide, CO2, is the end product from animal respiration and also combustion of organic substances.
The equation of animal respiration
C6H12O6 + O2 → 6CO2 + 6H2O
Combustion of ethanol fuel in a spirit burner
C2H5OH + 3O2 → 2CO2 + 3H2O
The periodic table
The periodic table displays all of the elements in a logical order according to their electronic configuration. This gives an arrangement where the metals are on the left and the non-metals are on the right.
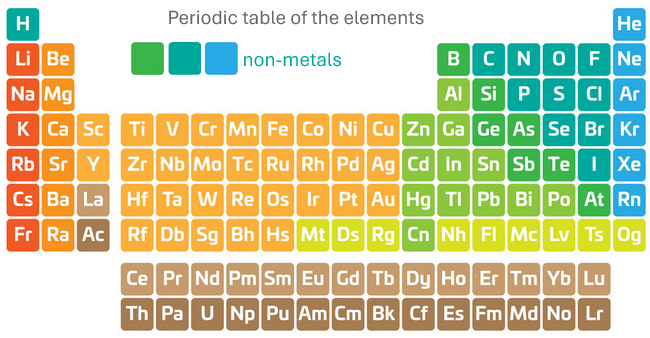
The properties of oxides of metals (metal oxides) are very different to the properties of non-metal oxides.
Metal oxides
- sodium oxide, white solid
- magnesium oxide, white solid
- calcium oxide, white solid
Non-metal oxides
- carbon dioxide, colourless gas
- nitrogen dioxide, red-brown gas
- sulfur dioxide, colourless gas
Apart from their obvious appearance, oxides of metals and non-metals also behave very differently when dissolved in water.
Non-metal oxides make acids when dissolved in water, whereas metal oxides form bases.
The reaction of sodium oxide with water
Na2O + H2O → 2NaOH
Sodium hydroxide, NaOH, is a very strong base.
The reaction of sulfur dioxide with water
SO2 + H2O → H2SO3
Sulfuric(IV) acid, H2SO3, is an acid.
Carbon dioxide
Carbon dioxide is a very important non-metallic oxide. It is involved in respiration in both animals and plants, and it is involved in the carbon cycle (see below). Carbon dioxide is a weakly acidic gas that dissolves in water forming a slightly acidic solution, called carbonic acid.
The oceans dissolve a large amount of the carbon dioxide in the air.
When gases dissolve in water, their solubility decreases as the temperature increases. Warming global ocean temperature releases more carbon dioxide into the air to the benefit of plants, but to the detriment of sea creatures, that need the carbon dioxide to construct their shells and bony growths.
Another major issue is that rising carbon dioxide levels increase the temperature of the earth via the greenhouse effect. This has many consequences.

The greenhouse effect
The Greenhouse effect was covered in MYP9 - this is just a summary.
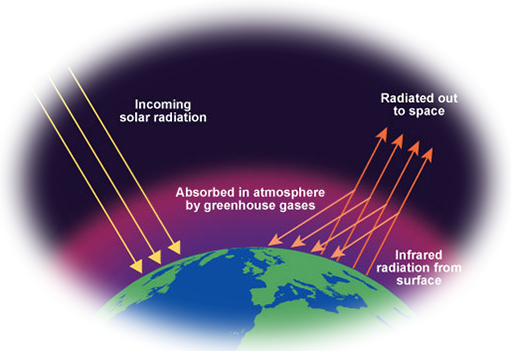
- Heat and light from the sun arrive at the surface of the Earth and heat it up.
- This heat is radiated back into the atmosphere, where some of it is absorbed by the greenhouse gases, water vapour, carbon dioxide and methane.
- If the concentration of these gases in the atmosphere increases, then the temperature of the Earth will increase and the world will experience global warming.
- Consequences: melting icecaps, rising sea-levels, loss of marine life, desertification, extreme weather events, destruction of habitats and extinction events.
Check your understanding
- What is the general difference between metal oxides and non-metal oxides?
- What happens when sulfur dioxide dissolves in water?
- Why is carbon dioxide considered a weakly acidic gas?
9.7 - Atmospheric pollution
Pollution refers to gases and microparticulates that have been produced in the air due to human activity and which are harmful to living creatures.
The main pollutants are:
- PM2.5 microparticulates
- Sulfur dioxide
- Nitrogen oxides
- Carbon monoxide
- Ozone (at ground level)
This is not intended as a complete list, but it shows the most important pollutants.
PM2.5 microparticulates
These are tiny particles of carbon produced from incomplete combustion, that measure up to 2.5 micrometres (microns) = 2.5 x 10-6 m in diameter. These particles are so small that they spend a long time in the air and get ingested into the body through normal breathing. Once in the lungs they are a focus for infection and a possible source of cancers. Microparticles are responsible for more deaths per year than any other type of pollution.
May countries have cleaned up their air compared to 50 years ago, but the problem of microparticles still exists.
Sulfur dioxide
This is a toxic gas produce by both volcanic activity (natural) and burning fossil fuels (anthropogenic). It has the chemical formula SO2. Sulfur dioxide is one of the pollutant gases responsible for acid deposition (rain, snow, dust) that can destroy forests and acidify lakes making them uninhabitable.
Nitrogen oxides
These are actually a group of chemicals all of which contain nitrogen and oxygen. They are given a general formula NOx. They are acidic gases that cause lung disease and contribute to other environmental problems.
Ozone
Ozone, O3 is an essential gas, but only in the upper stratosphere, where it protects the Earth from harmful ultraviolet light from the sun. At ground level it is a poisonous gas responsible for lung problems and illness. It is created by electric discharges, such as occur in internal combustion engines in cars.
Check your understanding
- List three major air pollutants and describe one harmful effect of each.
- Why are PM2.5 particles particularly dangerous to humans?
- What is the difference between ozone in the stratosphere and ozone at ground level?
9.8 - The carbon cycle

The carbon cycle explains how carbon moves around our ecosystem. It is used by animals in the form of carbohydrates for energy. It is used by plants in the form of carbon dioxide for energy and growth. It exists in the atmosphere and also dissolved in the oceans. It is tied up in rocks as inorganic carbonates, and as fossil fuels underground.
The whole ecosystem is balanced and has been for millions of years. It is only in the past couple of hundred years that human activity has been disturbing this balance, with devastating consequences for the ecosystem.
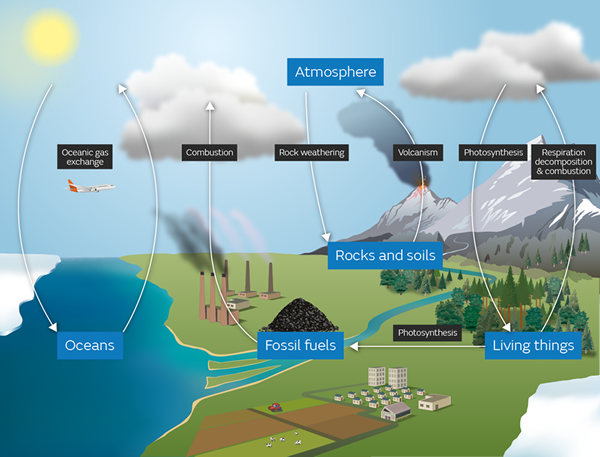
Check your understanding
- Describe two ways carbon dioxide enters the atmosphere.
- Explain how human activity is disturbing the natural carbon cycle.
- What role do oceans play in the carbon cycle?
9.9 - The rate of chemical reactions
Chemical reactions are processes that cause new substances to be formed. The rate of a chemical reaction is how fast this happens. Some reactions are very fast (think explosions) and some reactions take place over millions of years.
If we wish to measure how fast a chemical reaction happens it needs to be neither too fast nor too slow. We also have to decide what to measure. If we want to know how fast a car travels then we just need to measure how far it goes over a period of time.
To measure how far a chemical reaction has gone we need to have something visible that tells us how much of a reactant has been used up, or how much product has been formed.
Reactions that produce gases
Fortunately, there are some reactions that make this measurement easy because the reaction produced a gas. Gases are easy to collect and measure in gas syringes, or over water. We can simply measure the volume of gas produced per unit time.
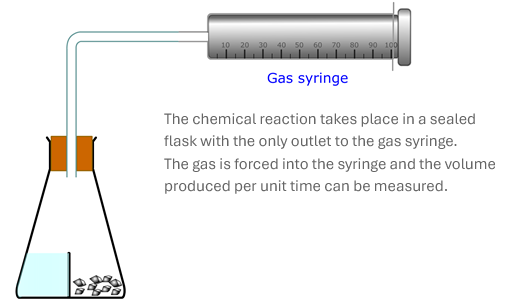
Reactions that produce dense gases
Reactions that produce carbon dioxide, which is a relatively dense gas can be monitored by measuring the total mass of the reactions vessel. As the carbon dioxide leaves the mass goes down.
It is possible to do the reaction in a container, such as a conical flask, on the top-pan electronic balance and record the mass.

Problem!
There is a problem measuring the rate of a chemical reaction in that the rate actually changes as the reactants are used up. This is because there are increasingly fewer reactant particles to collide.
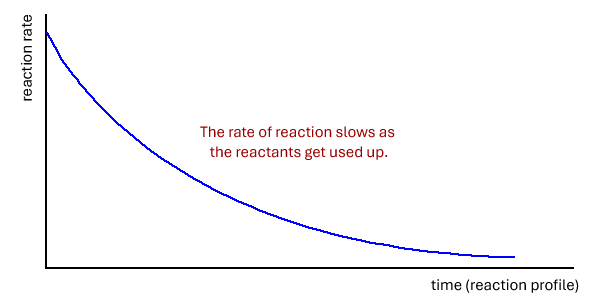
Chemical reactions are fastest (highest rate) at the beginning of a reaction and from there they get progressively slower.
This means that we must decide when to measure the rate. However, there is only one time that we actually know the concentration of the reactants, and that is at the beginning of the reaction. If we measure the rate at the very beginning it is called the initial rate. However, it is not always easy to determine the initial rate.
A suitable alternative is to measure the average rate over a pre-determined time-span of the reaction.
Finding the rate from graphs of gas produced against time
The definition of rate is change of amount of reactant or product/time
This is given as the gradient (slope) of the tangent to any point on the curve of mass loss, or volume of gas produced against time.
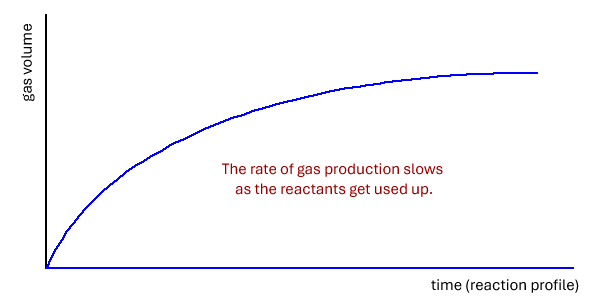
Tangent = a line that just touches a curve and is perpendicular to the radius of the curve at that point.
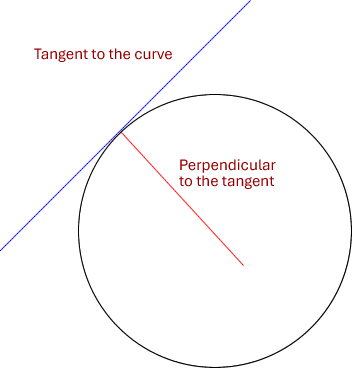
Factors affecting the rate of a chemical reaction
Changing the reaction "conditions" can affect how fast the reaction proceeds. There are several factors that can affect the rate of a chemical reaction:
- Temperature
- Concentration of reactants
- Surface area of reactants
- Catalysts
Temperature
Increasing the temperature at which a reaction happens increases the rate. The rule of thumb (approximate) is that the rate will double for every 10 °C increase. This is explained by the increased energy available for the colliding particles. The average collision is much more energetic and more likely to cause a reaction.
Concentration
Increasing the concentration of the reactants usually increases the rate. This is explained by the increase in the number of colliding particles. The number of collisions increases and therefore the reaction goes faster.
Surface area
Increasing the surface area of the substances reacting increases the rate. This only applies to solids in a reaction, as substances that are dissolved are already in a maximum total surface area condition. Collisions occur at the surface of the particles and the smaller the lumps of solid, the more surface is exposed to collision.
Catalysts
Some substances have an effect on the reaction rate without themselves seeming to take part in the reaction. This is an illusion, in that catalysts DO take part in the reaction, but they are regenerated during the overall process. A substance that increases the rate of reaction without being used up is called a catalyst, while a substance that slows a reaction down is called an inhibitor.
Check your understanding
- What is the definition of reaction rate?
- Why does an increase in temperature increase the rate of reaction?
- What is the difference between the initial rate and the average rate of a chemical reaction.
10u9.10 - Now test yourself
Click on the button below to access the self-tests for MYP9 and MYP10.