Unit 9 - Every breath you take
Content
Scheme of work
PS9.1 - Temperature and heat
Temperature scales
Celsius, Fahrenheit and Kelvin scales are used to measure temperature.
Heat refers to the ambient kinetic energy of the particles in the environment under study. Heat is experienced by the sensory cells, which pass information to the central nervous system regarding the vibrations of the particles.
Temperature scales give us a numerical value of the average kinetic energy of the particles.
The Kelvin scale is an absolute measure of the average kinetic energy in which the Kelvin value is directly proportional to the energy. Zero on the Kelvin scale means that there is no kinetic energy in the particles.
Interconversion of temperature scales
To convert Celsius to Kelvin you must add 273.
To convert Kelvin to Celsius you must subtract 273.
Convert 298 K to degrees Celsius
Degrees Celsius = Kelvin - 273
Celsius = 298 - 273 = 25ºC
PS9.2 - The atmosphere
The layers of air that we need to survive are known as the atmosphere. The solid land mass is called the lithosphere and the oceans and rivers, the hydrosphere. Together they make up our planet.
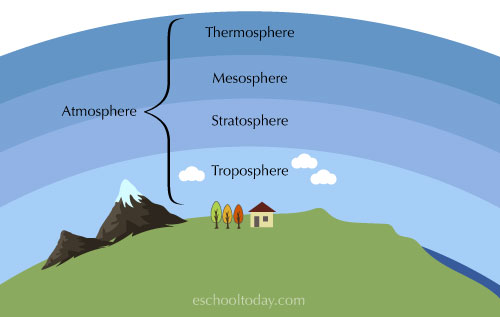
The lowest layer of the atmosphere is where all of the weather happens, it is known as the troposphere.
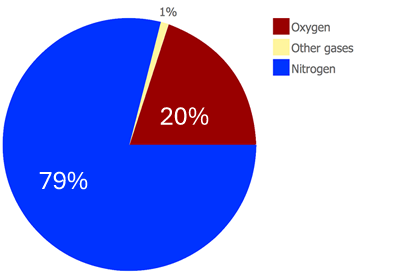
There are several gases in the atmosphere:
- Nitrogen, about 79%
- Oxygen, about 20%
- Argon, about 1%
- Carbon dioxide, between 0.02 and 0.04%
The problem is that every one of the 8 billion people on the earth rely on the air for survival and that there are many other substances that are released into the air that makes survival very difficult.
PS9.3 - Incomplete combustion & PM
Most energy resources are based on fossil fuels and rely on combustion. The hydrocarbons present in the fossil fuels burn in air to produce carbon dioxide and water vapour.
For example, natural gas (methane)
CH4 + 2O2 → CO2 + 2H2O
However, if the supply of air is limited then the combustion is incomplete and produces carbon monoxide and carbon micro-particulates (PM)
These carbon microparticulates are so small that they remain suspended in the air and become a major health hazard, causing bronchial complaints, asthma and cancer.
Most developed countries monitor these micro-particulates in cities and try to minimise their effect.
They are measured in μg per m3 air and determined according to the size of the microparticulates in micrometres (μm, 10-6 m).
Particulate matter with particle diameter up to 50 μm is known as PM50
PS9.4 - Pollution
Definitions
The terms "pollution" refers to anything in the environment that shouldn't be there and which has a detrimental effect, either in terms of health or environmental degradation.
In general, these substances are introduced into the environment as a result of human activity (anthropogenic).
Pollution of the air is particularly damaging, as everyone needs to breathe continuously introducing the pollution into the body. This has negative health consequences.
Air pollution is one of the world's leading risk factors for death. Air pollution is responsible for millions of deaths each year
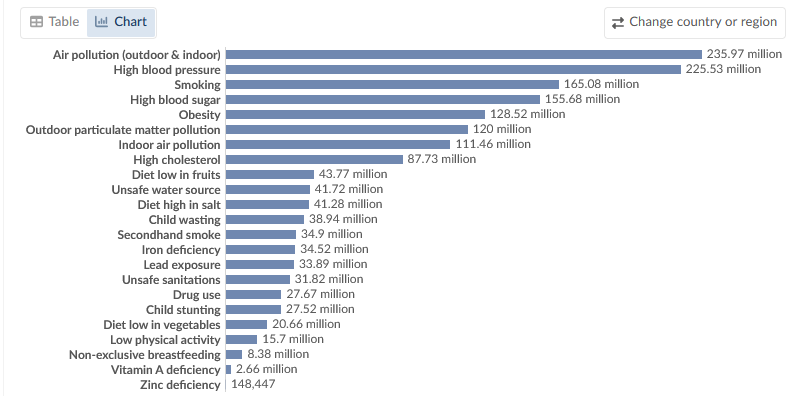
Our World in Data - Air pollution
Some pollutants also have natural sources. However, the balanced ecosystems of the Earth usually have mechanisms to reduce the impact of natural pollutants.
Significant air pollutants
- Sulfur dioxide, SO2
- Nitrogen oxides, NOx
- Microparticulates, PM2.5
Sulfur dioxide, SO2
Sulfur dioxide is an acidic gas with a suffocating metallic smell (and taste).
It reacts with water making sulfuric(IV) acid, a weak acid:
The reaction of sulfur dioxide with water
SO2 + H2O → H2SO3
Anthropogenic sources
Sulfur is an element that appears in the protein building blocks of life, the amino acids methionine and cysteine. As fossil fuels are derived from living organisms, they contain sulfur. When fossil fuels are burned the sulfur content turns to sulfur dioxide.
Any industry or activity that burns fossil fuels produces sulfur dioxide. Energy generation (oil or coal fired power stations) and traffic are the main activities.
Natural sources
Sulfur dioxide also has a natural source in volcanic activity.
The reaction of sulfur with oxygen
S + O2 → SO2
Health effects
Sulfur dioxide creates sulfuric acid in the lungs if inhaled. This can lead to all sorts of health issues.
The reaction of sulfur dioxide with water
SO2 + H2O → H2SO3
Aerial oxidation of sulfuric(IV) acid to sulfuric(VI) acid
2H2SO3 + O2 → 2H2SO4
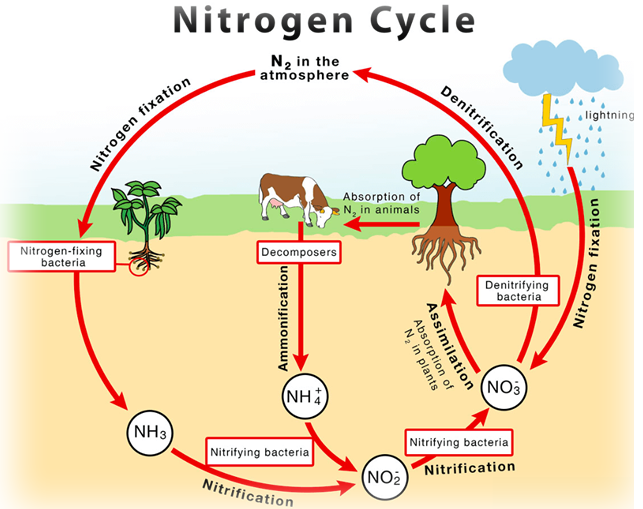
Nitrogen oxides
These are formed naturally by electrical discharges in the air (lightning), and are part of the natural nitrogen cycle, which is essential for plant life.
Nitrogen oxides are made by electrical discharges:
N2(g) + O2(g) → 2NO(g)
Nitrogen(II) oxide reacts with oxygen in the air:
2NO(g) + O2(g) → 2NO2(g)
The nitrogen(IV) oxide, NO2, dissolves in rainwater and gets into the ground where it is converted into soluble nitrates in the soil. The plants extract these soluble nitrates through the roots and use the nitrogen for growth. When the plants die the nitrogen is returned to the atmosphere as ammonia, where it is oxidised to nitrogen oxides again.
Microparticulates
Also known as particulate matter (PM).
The mass of particulate matter with maximum diameter of particles is defined. These are fundamentally small particles of carbon (sometimes called black carbon) produced during incomplete combustion.
PM2.5 means microparticulates with diameter up to 2.5μm = 2.5 x 10-6 m
These microparticulates remain in the air for long periods of time due to their small size and get inhaled by the population. They are responsible for respiratory diseases and lung cancers.
Activity
Prepare a powerpoint presentation on one type of air pollution using the World in Data website and any other resources. The presentation should have only 5 slides.
PS9.5 - Gases
As a state of matter, gases present specific difficulties when it comes to their collection and containment.
Properties of gases - volume
A substance is gaseous if its particles have enough energy to overcome the forces of attraction between them. For this reason gases expand to fill all of the available volume.
Properties of gases - temperature
The volume and pressure of a gas is directly proportional to its absolute temperature. As the temperature increases, so does the volume (at constant pressure) or the pressure (at constant volume).
The dependence of pressure on temperature
P ∝ T (at constant volume)
The dependence of volume on temperature
V ∝ T (at constant pressure)
At constant temperature, the product of the pressure and the volume is constant.
The relationship between volume and pressure at constant temperature
PV = constant (at constant temperature)
Properties of gases - collection
The preparation and gas collection apparatus must be gas-tight or the gas must be prevented from leaving the collection apparatus in some way.
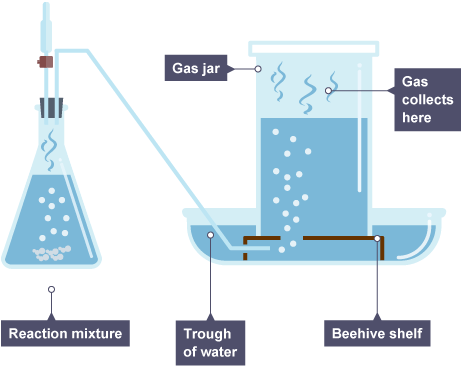
This would be suitable for hydrogen, oxygen and carbon dioxide gases.
Gases such as ammonia, hydrogen chloride and sulfur dioxide are too soluble in water and must be collected in another way.